Validating European patents in Cambodia
Download
Validation of a European patent means the recognition, on request, of the effects of the European patent application and the European patent in Cambodia. We provide below some critical remarks on the matter for your reference.
| No. | Titles | Remarks |
| 1 | Validation Agreement | - On 23 January 2017, the Agreement on Validation of European Patents (the “Validation Agreement”) was signed between the European Patent Office (“EPO”) and the Royal Government of the Kingdom of Cambodia.
- The Validation Agreement was ratified under Royal Kram No. NS/RKM/1117/017 dated 24 November 2017 on the Ratification of the Agreement on Validation of European Patents.
- The validation procedures were set forth in Prakas (declaration) No. 282 MIH/2017 dated 8 December 2017 on the Regulation and Procedures for the Validation of European Patents in Cambodia.
|
| 2 | Effective date | - On 1 March 2018, the Validation Agreement came into force, making Cambodia the first Asian country to recognize the validity of the patents granted by the EPO on its territory.
|
| 3 | Benefits | - The entry into force of the Validation Agreement with Cambodia enables European patents to be validated in Cambodia, thereby extending their rights to Cambodia. To this end, the EU patent holders need to file requests for validation of their European patents in Cambodia with no requirement to file a national patent application in the country. Such validation is available for both applications directly filed with the EPO, as well as European PCT applications.
- European patents granted by the EPO and validated in Cambodia will have the same legal effects as a corresponding Cambodian patent and will be subject to Cambodian patent law.
- As compared to filing of a national patent application, the validation may save some kinds of fees and accelerate the grant of a patent in Cambodia.
|
| 4 | Requirements | The following requirements must be met to validate a European patent in Cambodia: - The European patent must have a filing date on or after 1 March 2018. It is not available for applications filed prior to that date, or for any European patents resulting from such applications.
- Required documents:
(i) Notarized Power of Attorney; (ii) Granted European patents (notarization/certification is required for the copy of granted European patents); (iii) Copy of the granted patent specification of the European patent in English (notarization/certification is not required for such copy). - The technical solutions under the European patents must meet the Cambodian requirements for the patent protected subject matters.
|
| 5 | Exclusions | - Per Article 136 of the Cambodian Law on Patents, Utility Models and Industrial Designs, pharmaceutical products are excluded from patent protection. Cambodia currently benefits from the World Trade Organization waiver that allows Least Developed Countries (LDCs) to avoid granting and enforcing IP rights on pharmaceutical products until 2033. This waiver would also apply to European patents that provide protection for pharmaceutical products, for which validation is sought in Cambodia.
- Applicants can, however, benefit a so-called “mailbox system” under Article 70.8 TRIPS. According to this “mailbox system”, Cambodian legislation authorizes the filing of patent applications for pharmaceutical products, despite the fact that they are excluded from patent protection. These national applications will not be examined as to patentability until the transitional period is over. Following that period, protection may be granted for the remainder of the patent term, calculated from the filing date of the application.
|
| 6 | Flowchart for validation | Download 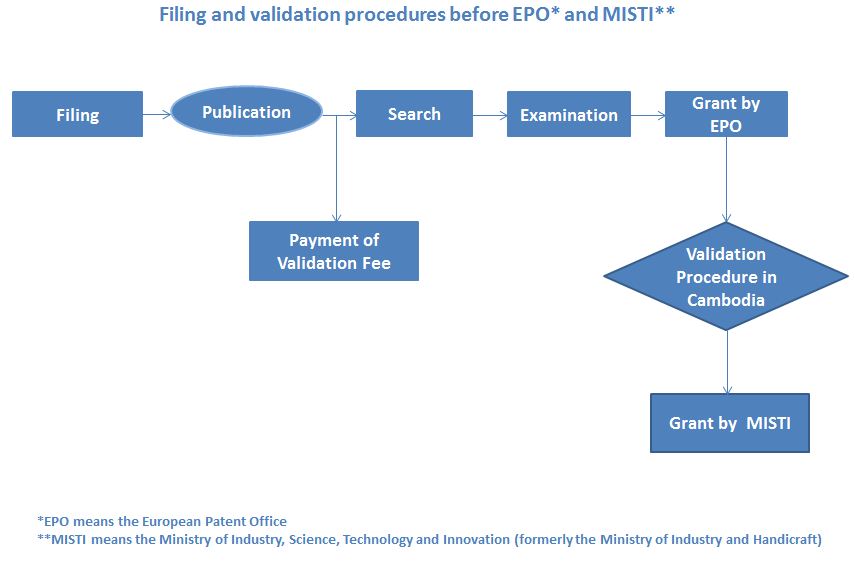
Note: – As mentioned above, pharmaceutical product patents are excluded from protection under Cambodian patent law, and therefore cannot be validated. – Once the validation is successful, the patent owner will be granted an official patent certificate by MISTI. |
| 7 | Others | - The applicant shall file their application and pay any associated fees to EPO by themselves.
- For the time being, upon validation of European patents in Cambodia, fees for granting patent, publication, translation and annuity fee shall be incurred.
- To date, MISTI has not issued any form for filing validation of European patents on demand in Cambodia. When the form is issued by MISTI, there may be other fees incurred for filing such validation in Cambodia.
|
Related Articles:


